Phase 1 trial shows U of G vaccine against global cause of bacterial diarrhea is safe and efficacious
University of Guelph researchers are one step closer to finalizing the first human vaccine for Campylobacter jejuni, a bacterial species found in raw or undercooked chicken that causes major diarrheal illness worldwide.

New results from a Phase 1 human trial showed that the vaccine was safe and efficacious in raising antibodies for C. jejuni in humans. The main objective of a Phase 1 trial is to determine whether a vaccine is safe.
“This human clinical trial hit the mark,” says Dr. Mario Monteiro, professor in the Department of Chemistry.
Monteiro led the development of the vaccine in the College of Computational, Mathematical and Physical Sciences while working with U.S. partners.
Unique in the world for developing sugar-based vaccines, his lab discovers sugar antigens and makes them immunogenic for use as vaccines.
The randomized, double-blinded trial for the vaccine took place between 2022 and 2024 at the Cincinnati Children’s Hospital Medical Centre. Three doses of the vaccine known as Campylobacter jejuni Conjugate Vaccine (CJCV2) were given to about 60 healthy adults at various doses.
Funded by the U.S. National Institutes of Health, the trial showed adverse effects were minimal and mostly associated with symptoms at the injection site: muscle pain, tenderness and fatigue.
“The exciting part is that the highest response was against a low dose of the vaccine,” Monteiro says.
This makes the results all the more promising for a multivalent product, a vaccine that could cover multiple C. jejuni strains at once.
“It is a historic milestone for U of G and Canada’s vaccine research and innovation landscape.”
Campylobacter affects millions, vaccine could help all ages
One of the top bacterial causes of diarrheal illness worldwide, C. jejuni can cause severe diarrhea, fever and cramps usually lasting for several days. Left untreated, however, cases can be fatal. An outbreak due to contaminated water last year killed 11 people in India.
Many in the developing world are uniquely at risk from the infection, including children, as well as those with weakened immune systems.
“Travellers of any age will benefit from this vaccine,” Monteiro says. “The vaccine will also indirectly prevent the onset of the severe autoimmune disorder, Guillain-Barré syndrome, which usually shows up after a C. jejuni infection.”
Vaccine shows power of U.S. and Canada collaboration
More than 20 years in the making, the CJCV2 vaccine was developed in collaboration with Canadian researchers and the Naval Medical Research Center (NMRC), which tested the vaccines made at U of G.
During development, the vaccine was recognized as a preferred product by the World Health Organization for potential use against what medical experts call a “hidden pandemic,” a growing resistance to antibiotics, as well as the WHO’s priority pathogens list.
“Our first human trial in 2014 also showed safety, but it did not hit the mark of being efficacious in mounting an immune response,” says Monteiro. “We found out that an important part of the polysaccharide [sugar] was missing in the first vaccine.”
This time, he says, his lab confirmed that a key region of the molecule known as an epitope was present. The result was a vaccine that was efficacious in raising antibodies against the target.
U of G lab unique for making sugar-based vaccines
The study marks the fourth human trial for vaccines based on discoveries by Monteiro – three for C. jejuni and one for C. difficile.
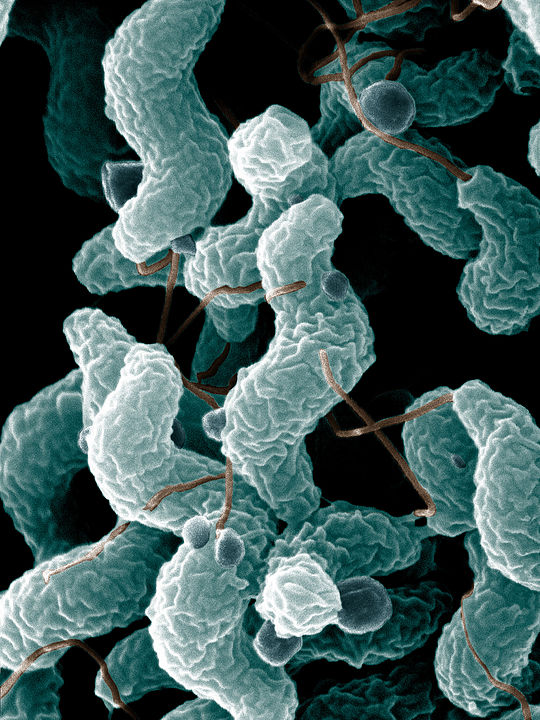
Many vaccines focus on targeting the proteins of a pathogen. Monteiro’s vaccines, however, help the immune system recognize specific carbohydrates, or sugars, on the surface of a bacterial cell. This makes them distinct from gene-based vaccines as well, like the ones developed for COVID-19.
Carbohydrate-based vaccines are relatively unexplored because they involve complex chemistry, making development extra challenging and time-consuming, Monteiro says. However, for bacteria like Campylobacter, rich in sugar structures, they can be more effective in inducing an immune response.
Championing the research was Dr. David Hobson, manager of the Research Innovation Office (RIO) at U of G. RIO guides faculty through processes of marketing and commercialization, connecting researchers with industry partners to turn research into real-world solutions.
“RIO has always worked collaboratively with the NMRC to support industry partners that have the resources to commercialize these vaccines globally,” says Hobson. “These vaccines have tremendous potential. We are especially interested in helping the industry get them to market.”
A Phase 2 trial is next, Monteiro says.
This will determine if the immune response observed can prevent diarrheal illness, testing against a placebo with hundreds more volunteers.
“Commercialization of science is on everyone’s minds these days,” Monteiro says, “but the marketability is not our main motivation. My students and I simply like to discover new molecules every day.
“But when it reaches the clinical shelves, it could save a lot of lives.”
